Greetings!
It appears the FDA is moving quickly. As of today 23andMe can no longer sell DNA kits for health purposes. Perhaps this will change if things are resolved with the FDA, but that could take much time.
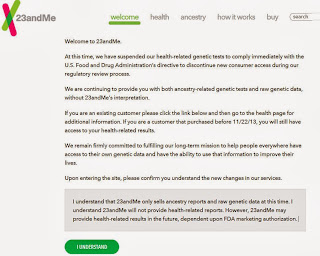
When you first open 23andMe the following message appears. You must click that you understand the information before you can actually login. This appears only once.
The above form letter states:
Welcome to 23andMe.
At this time, we have suspended our health-related genetic tests to comply immediately with the U.S. Food and Drug Administration’s directive to discontinue new consumer access during our regulatory review process.
We are continuing to provide you with both ancestry-related genetic tests and raw genetic data, without 23andMe’s interpretation.
If you are an existing customer please click the link below and then go to the health page for additional information. If you are a customer that purchased before 11/22/13, you will still have access to your health-related results.
We remain firmly committed to fulfilling our long-term mission to help people everywhere have access to their own genetic data and have the ability to use that information to improve their lives.
Upon entering the site, please confirm you understand the new changes in our services.
I understand that 23andMe only sells ancestry reports and raw genetic data at this time. I understand 23andMe will not provide health-related reports. However, 23andMe may provide health-related results in the future, dependent upon FDA marketing authorization.
I UNDERSTAND
After clicking the green button below the message (see photo above) which says "I UNDERSTAND", you are taken to the home page for selling kits. The kit price is still $99, but there is no mention of any health testing.
IF the FDA's issues with 23andMe can be resolved and the company can resume selling DNA health kits, the path would be cleared for other companies. However, if 23andMe either has to cease selling the health tests or has to modify their system greatly, there is concern about how profitable the company will be, given that it has always been structured around a focus on health rather than on genealogy. In the meantime, it is also possible that more restrictions may be placed upon the company. We shall have to see.
Depending upon the outcome, their entire business model may need changing and/or their website. No doubt the FDA has 23andMe's ear now, and no doubt 23andMe will scramble to comply. They have no choice. BUT just what will be required of them is the question What compromise, if any, can be reached.
Regardless, this can be a handicap to genealogists. Many genetic genealogists have reported that even though people have tested for health reason, they often become interested in genealogy. Given this, all of us are losing.
The genetic genealogy community is watching all this very carefully...
Emily
It appears the FDA is moving quickly. As of today 23andMe can no longer sell DNA kits for health purposes. Perhaps this will change if things are resolved with the FDA, but that could take much time.
When you first open 23andMe the following message appears. You must click that you understand the information before you can actually login. This appears only once.
The above form letter states:
Welcome to 23andMe.
At this time, we have suspended our health-related genetic tests to comply immediately with the U.S. Food and Drug Administration’s directive to discontinue new consumer access during our regulatory review process.
We are continuing to provide you with both ancestry-related genetic tests and raw genetic data, without 23andMe’s interpretation.
If you are an existing customer please click the link below and then go to the health page for additional information. If you are a customer that purchased before 11/22/13, you will still have access to your health-related results.
We remain firmly committed to fulfilling our long-term mission to help people everywhere have access to their own genetic data and have the ability to use that information to improve their lives.
Upon entering the site, please confirm you understand the new changes in our services.
I understand that 23andMe only sells ancestry reports and raw genetic data at this time. I understand 23andMe will not provide health-related reports. However, 23andMe may provide health-related results in the future, dependent upon FDA marketing authorization.
I UNDERSTAND
After clicking the green button below the message (see photo above) which says "I UNDERSTAND", you are taken to the home page for selling kits. The kit price is still $99, but there is no mention of any health testing.
IF the FDA's issues with 23andMe can be resolved and the company can resume selling DNA health kits, the path would be cleared for other companies. However, if 23andMe either has to cease selling the health tests or has to modify their system greatly, there is concern about how profitable the company will be, given that it has always been structured around a focus on health rather than on genealogy. In the meantime, it is also possible that more restrictions may be placed upon the company. We shall have to see.
Depending upon the outcome, their entire business model may need changing and/or their website. No doubt the FDA has 23andMe's ear now, and no doubt 23andMe will scramble to comply. They have no choice. BUT just what will be required of them is the question What compromise, if any, can be reached.
Regardless, this can be a handicap to genealogists. Many genetic genealogists have reported that even though people have tested for health reason, they often become interested in genealogy. Given this, all of us are losing.
The genetic genealogy community is watching all this very carefully...
Emily
No comments:
Post a Comment